연구배경
국내에서 건강기능식품 및 의약품으로 사용되고 있는 글루코사민은 현재(2011년 9월)
35개(황산염 글루코사민)의 제품이 의약품으로 허가되어 있으며(식약청, 2010), 건강보험
심사평가원(2009)자료에 따르면 황산염 글루코사민의 건강보험 EDI(Electronic Data Interchange) 청구액은 연간 최소 80억원에 이르는 것으로 추정된다. 그러나, 국외의 경우, 이탈리아, 영국(이상 전문의약품), 독일, 한국, 대만, 태국 등(이상 일반의약품)에서 의약품으로 허가되어있으나, 급여로 인정되는 국가는 한국, 대만, 태국 등 에 불과하다. 게다가, 국내에서는 별도의 기준 없이 식약청 허가사항에 준해 급여하고 있으나, 대만에서는 엄격한 기준에 의해 급여되고 있으며 태국에서는 최근 보험급여에서 제외하려는 시도중에 있다.
한국보건의료연구원에서는 체계적 문헌고찰 및 메타분석을 통해 2009년 12월 ‘골관절
염 환자에서 글루코사민과 콘드로이틴의 효과’ 임상적 효과에 대해 근거 없음을 발표한바
있으며, Wandel 등(2010)의 연구에서도 임상적 관점에서 인정할 만한 효과는 없다고 발
표한바 있다. 그럼에도 불구하고 글루코사민은 여전히 건강기능식품뿐 아니라 의약품으로
인정되고 있으며, ‘경증에서 중등도의 골관절염’환자를 대상으로 성인의 경우 1회 500mg
을 1일 3회 6주간 복용할 경우 건강보험에서 급여로 인정되고 있어, 건강보험급여의 적절
성 여부 및 소비자 혼란 등 사회적 논란이 계속되고 있다.
이에, 한국보건의료연구원에서는 기존 보고서인 '골관절염 환자에서 글루코사민과 콘드로이틴의 효과’(한국보건의료연구원, 2009)의 후속연구로 글루코사민의 임상적 효과에 대한
현재근거를 재평가하였다.
연구방법
본 연구는 한국보건의료연구원의 신속근거평가프로그램인 RAPID를 통해 신속 체계적문
헌고찰방법으로 수행되었으며, 동 연구원의 2009년 보고서와 기존 보고서 이후 출판된 문
헌을 토대로 검토하였다. 기존 보고서와 문헌검색데이터베이스, 검색전략, 선택배제기준, 자
료추출형식등은 기존보고서와 동일하게 따랐으며, 현존하는 문헌검토를 위해 Ovidmedline,
Embase, Cochrane library(Cochrane systematic reviews database, DARE, NHS EED, HTA database포함), CRD database, Pubmed, CCTR, koreamed를 이용하여 2011년 8월 22~23일 검색을 완료하였다. 선택기준은 골관절염환자에서 글루코사민의 임상적 효과성을 보고한 무작위임상시험연구와 체계적 문헌고찰 연구로 하였으며, 문헌선택과정과 자료추출 및 질평가는 2명의 연구자에 의해 독립적으로 수행된 후 토의하여 합의를 이루었다.
연구결과
기존 보고서 이후 추가된 무작위임상시험연구는 총 2편(Chopra et al 2011; Sawitzke2011)이었으나, Chopra 등(2011)의 연구는 결과값이 그래프로 제시되어 있어 표준편차를 추출할 수 없어 기존 보고서와 통합한 추가메타분석에는 Sawitzke 등(2011)의 연구만 추가하였다. 추가메타분석을 통한 글루코사민의 임상적 효과를 의료결과별로 기술하면 다음과 같다.
통증감소효과- 글루코사민의 임상적 효과(염산염 글루코사민 + 황산염 글루코사민)
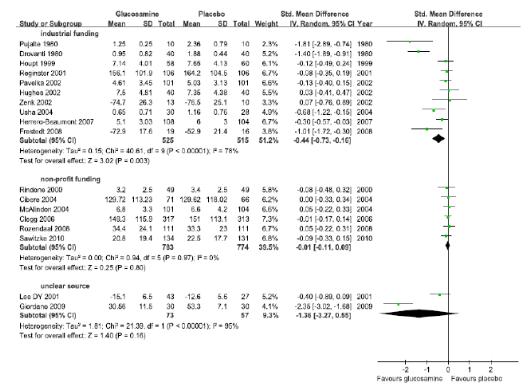
연구비출처에 따른 하위집단분석 결과, industrial funding을 받아 수행된 연구(10편)에 서 글루코사민 통증감소효과의 통합 추정치는 SMD -0.44(95% CI -0.73, -0.16)로 위약에 비해 효과적이었으나, I2 = 78%로 이질적이였고, non-profit funding을 받아 수행 된 연구(6편)는 SMD -0.01(95% CI -0.11, 0.09), I2 = 0%로 위약과 효과 차이가 없었다.
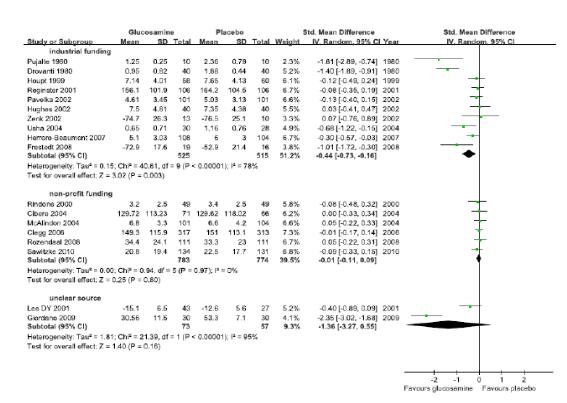
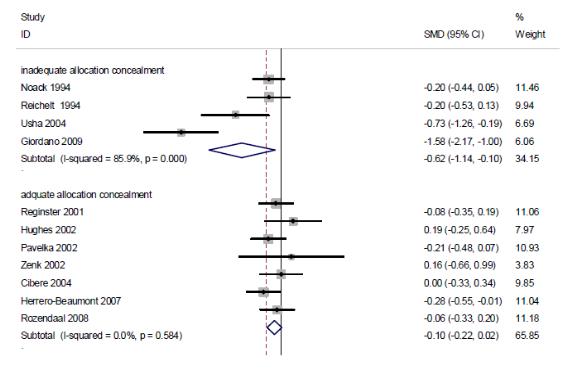
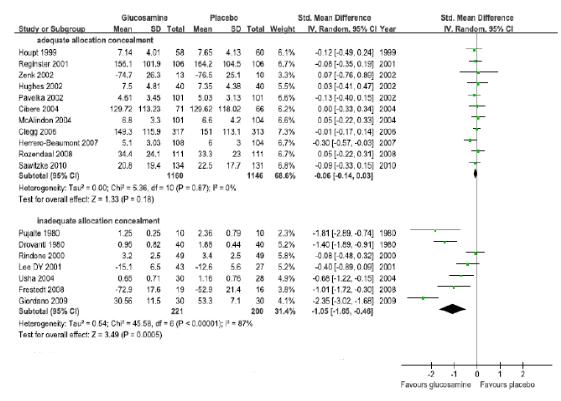
연구수행의 방법론적 질(배정은폐의 적절성)에 따른 하위집단분석결과, 배정은폐가 적절히 수행된 연구(11편)의 통합추정치는 SMD -0.06(95% CI -0.14, 0.03), I2 = 0%로 효과 없었으며, 이에 반해 배정은폐가 적절히 수행되지 않은 연구(7편)는 SMD -1.05(95% CI-1.65, -0.46)로 위약과 비하여 효과적이었으나, I2 = 87%로 이질적이었다.
- 황산염 글루코사민의 임상적 효과
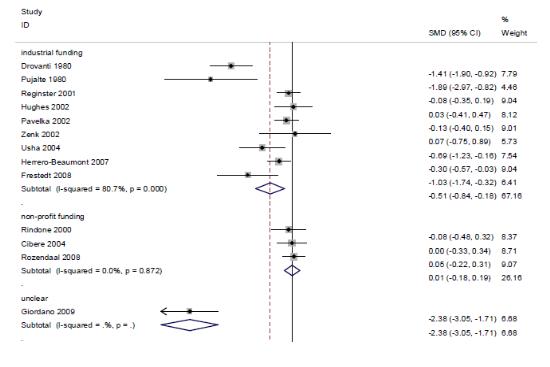
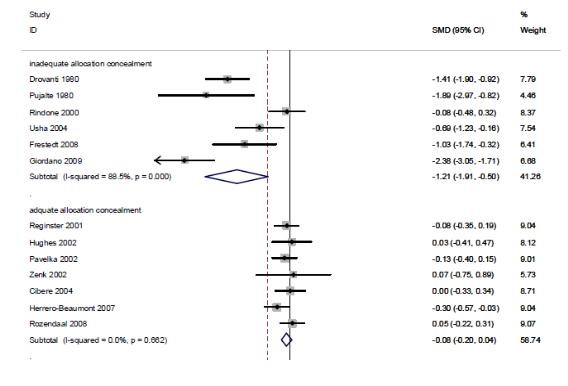
황산염 글루코사민의 임상적 효과만을 분석한 결과는 기존 보고서 이후 추가된 문헌이 없었기 때문에 기존 보고서 결과와 동일하며 다음과 같다. 연구비출처에 따른 하위집단분석 결과, industrial funding을 받아 수행된 연구(9편)의 통합 추정치는 SMD -0.51(95% CI -0.84, -0.18)로 위약에 비해 효과적이었지만, I2 =80.7%로 이질성을 보였고, non-profit funding을 받아 수행된 연구(3편)는 SMD 0.01(95% CI -0.18, 0.19)로 위약과 효과 차이가 없었으며, I2 = 0%였다.
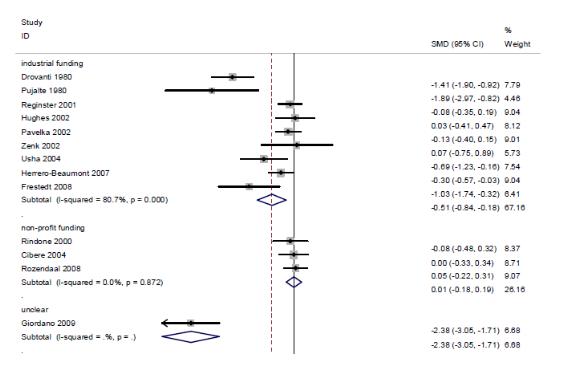
연구수행의 방법론적 질(배정은폐의 적절성)에 따른 하위집단분석을 수행한 결과, 배정은폐가 적절히 수행된 연구(7편)의 통증감소효과는 통합 추정치 SMD -0.08(95% CI-0.20, 0.04)로 위약과 효과 차이가 없었고, I2 = 0%였다. 이에 반해 배정은폐가 적절히 수행되지 않은 연구(6편)의 SMD -1.21(95% CI -1.91, -0.50)로 위약과 비하여 통증감소에 효과적이었으나, I2 = 88.5%로 이질적이었다.
관절기능향상효과
- 글루코사민의 임상적 효과(염산염 글루코사민 + 황산염 글루코사민)
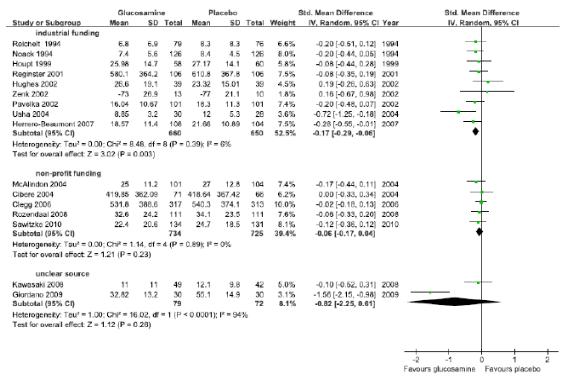
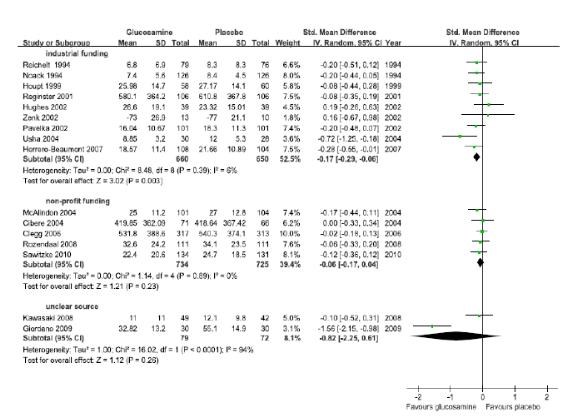
연구비출처에 따라 하위집단분석을 수행한 결과, Industrial funding을 받아 수행된 연구(9편)의 글루코사민의 관절기능 향상 효과에 대한 통합 추정치는 SMD -0.17(95% CI-0.29, -0.06), I2 = 6%로 위약에 비해 효과적이었으나, non-profit funding을 받아 수행된 연구(5편)의 SMD -0.06(95% CI -0.17, 0.04), I2 = 0%로 위약과 효과 차이가 없었다.
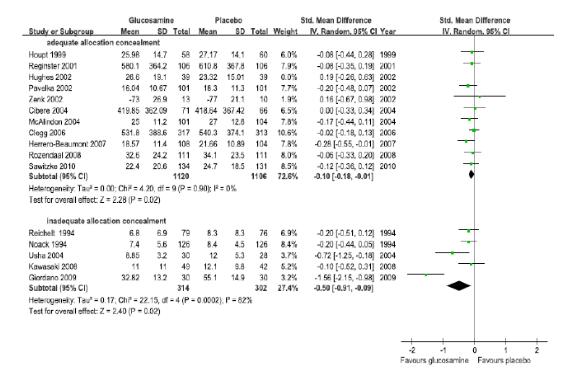
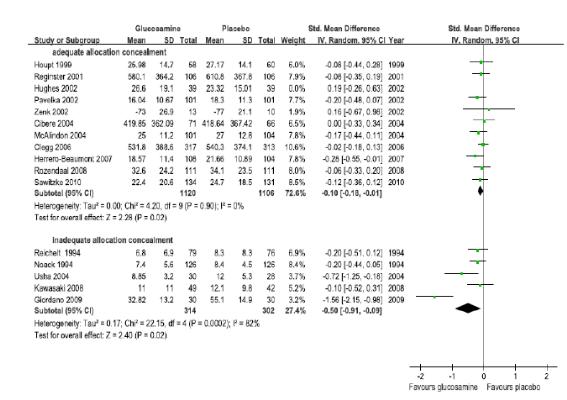
연구수행의 방법론적 질(배정은폐의 적절성)에 따른 하위집단분석을 수행한 결과, 배정은폐가 적절히 수행된 연구(11편)의 글루코사민의 관절기능향상 효과에 대한 통합추정치는
SMD -0.10(95% CI -0.18, -0.01), I2 = 0%로 통계적으로 매우 작은 효과를 보였으나, 대부분 임상적으로는 effect size가 0.5이상 되어야 의미를 두게 되므로 임상적인 큰 의미는 없는 차이로 판단된다. 또한, 배정은폐가 적절히 수행되지 않은 연구(7편)는 SMD -0.50(95% CI -0.91, -0.09)로 위약과 비하여 기능향상에 효과적이었으나, I2 = 82%로 이질적이었다.
- 황산염 글루코사민의 임상적 효과
황산염 글루코사민의 임상적 효과만을 분석한 결과는 기존 보고서 이후 추가된 문헌이
없었기 때문에 기존 보고서 결과와 동일하며 다음과 같다.
연구비출처에 따른 하위집단분석 결과, industrial funding을 받아 수행된 연구(8편)의 통합 추정치는 SMD -0.18(95% CI -0.30, -0.07)로 위약에 비해 효과적이었고 I2 =16.7%였다. 또한, non-profit funding을 받아 수행된 연구(2편)는 SMD -0.04(95% CI -0.24, 0.17)로 위약과 효과 차이가 없었으며, I2 = 0%였다.
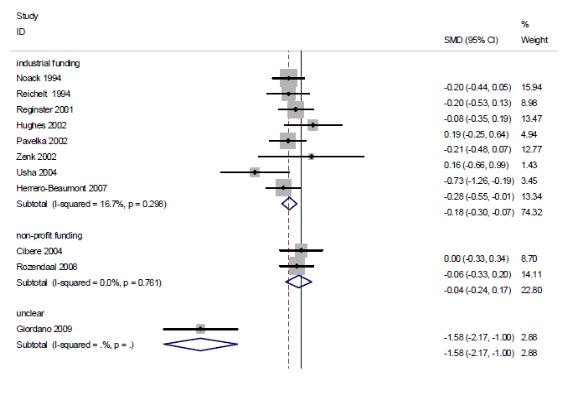
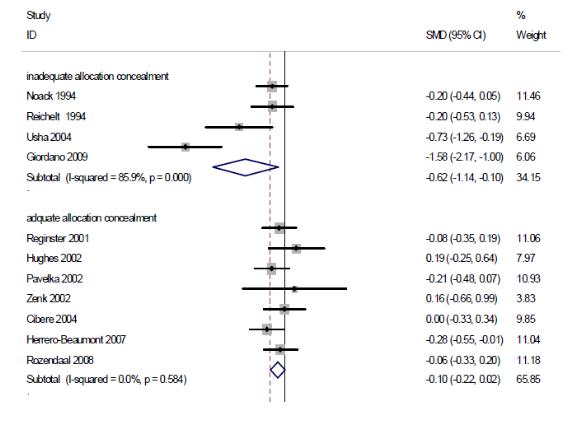
연구수행의 방법론적 질(배정은폐의 적절성)에 따른 하위집단분석을 수행한 결과, 배정은폐가 적절히 수행된 연구(7편)의 통합 추정치는 SMD -0.10(95% CI -0.22, 0.02), I2
= 0% 로 위약과 효과 차이가 없었고, 이에 반해 배정은폐가 적절히 수행되지 않은 연구
(4편)의 SMD는 -0.62(95% CI -1.14, -0.10)로 위약과 비교하여 기능 향상에 효과적이었으나, I2 = 85.9%로 이질적이었다.
결론
골관절염환자에서 글루코사민의 임상적 효과는 기존 보고서 및 추가 문헌을 통해 분석해 본 결과 위약과 비교하여 통증 감소, 관절기능 향상에 효과가 있는 것으로 나타났으나 결과에 일관성이 없었다. 하위집단분석시 글루코사민 제조사에 의해 연구비를 지원받지 않은 연구에서는 통증 감소와 기능 향상에 일관되게 효과가 없었다. 또한, 연구시작시점에 연구자로 하여금 연구 참여자가 어떤 치료그룹에 할당되었는지 모르게 이루어진 배정은폐가 적절했던 연구에서 글루코사민은 통증감소 효과가 없었으며, 관절기능향상에서는 통계적으로 매우 작은 효과(SMD -0.10(95% CI -0.18, -0.01) I2 = 0%를 보였으나 effect size가
0.5미만으로 임상적으로 의미있는 효과라고 보기는 어려웠다.
특히 국내에서 의약품으로 허가되어 급여로 인정되고 있는 황산염 글루코사민의 경우 임상적효과를 검토한 결과, 기존보고서 외에 추가된 문헌이 없었으므로 기존 보고서 결과를
그대로 인용하면 다음과 같다. 황산염 글루코사민은 위약과 비교시 제조사에 의해 연구비를
지원받지 않은 연구, 배정은폐가 적절히 이루어진 연구에서는 통증감소, 관절기능향상에 일관적으로 효과차이가 없었다.
본 연구보고서는 한국보건의료연구원의 RAPID program을 통해 짧은 시간내 신속하게
검토되어졌으므로 엄격성면에서 다소 부족한 부분이 있을 수 있으며 그로 인한 비뚤림 위험을 배제할 수 없다. 추후 필요시 완전한 체계적 문헌고찰 혹은 기존의 체계적 문헌고찰의 활용한 체계적 문헌고찰(using existing systematic review)에 의해 보완되어질수 있을 것이다.
제언
본 연구에서는 여전히 논란중에 있는 글루코사민의 임상적 효과를 재평가하여 현재 문헌적 근거를 종합하였으며, 배정은폐가 적절한 연구, 제조사로부터의 연구비지원이 없는 연구에서 글루코사민은 통증 및 관절기능향상에 있어 임상적으로 의미있는 효과가 없었다.
국내에서는 글루코사민이 건강기능식품뿐 아니라, 급여 일반의약품으로 인정되고 있으나,전 세계적으로 급여로 인정하고 있는 국가는 한국, 대만, 태국에 불과한 상황이다. 이는 건강보험재정에 부적절한 영향을 미칠수 있으며 임상적으로 충분하지 않은 효과를 위해 건강보험재정이 지원되어야 하는지는 추가적인 정책적 판단이 필요할 것으로 사료된다.
이러한 정책적 판단의 객관적 근거는 관련이슈와 이해관계가 없는 중립적인 연구기관에서의 연구결과가 토대가 될수 있다. 국외의 경우, 영국 NICE의 HTA 평가 결과가 3개월 내 NHS(National Health Services)에서 반영하도록 명문화되어 있으며, 이밖에 독일,호주, 캐나다, 태국, 스웨덴 등의 유사기관에서도 제도적으로 연구 결과물을 반영하는 장치가 마련되어 있다(부록 7.7).
지난 2009년 한국보건의료연구원에서 발표한 글루코사민의 효과와 관련된 연구는 이런
취지로 시행되었었으나, 연구결과가 행정적인 부분과의 연결이 잘 이루어지지 않았었다. 물
론 실제 시장에서의 매출 감소 등의 영향은 있었지만, 판매중지, 비급여로의 전환 등과 같은 정책적 결정까지 미치지는 못했다. 따라서, 국내에서도 이러한 중립적인 연구기관의 연
구결과를 사후 어떻게 정책에 반영할지 명문화하는 제도적 장치가 필요하다고 판단된다.
Research Background
Glucosamine is being used as functional health food and medical drug, and total of 35 products (glucosamine sulfate) have been approved as medical drugs in Korea (Korea Food & Drug Administration, 2010). According to the data from Health Insurance Review & Assessment Service (2009), the value of quantities of glucosamine sulfate produced annually is 14.2 Billion Korean Won and the health insurance EDI (Electronic Data Interchange) claim value is approximately 8 Billion Korean Won, However, in the case of overseas countries, only Italy and England approved glucosamine sulfate as ethical drug while Germany, Korea, Taiwan and Thailand,
etc approved it as over the counter. Only Korea, Taiwan and Thailand recognize it for health insurance payment. Moreover, while Korea is making payment for glucosamine sulfate based on the approvals of Korea Food & Drug Administration without separate standard in Korea, Taiwan makes payment in accordance with strict standard and Thailand is recently attempting to exclude it from insurance payment.
The National Evidence-based Health Care Collaborating Agency has announced that there is no basis for the clinical effect on ‘the effect of glucosamine and chondroitin on osteoarthritis in December 2009 through systematic review and meta-analysis, and the study by Wandel et al (2010) also announced that there is no effect of glucosamine that could be acknowledged from the clinical perspective. In spite of these findings, glucosamine continues to be recognized not only as functional health food but also as medical drug, and social disputes on glucosamine including the adequacy of health insurance payment and consumer confusion continue since payment by the health insurance is acknowledged if it is being administered 3 times
per day for more than 6 weeks with single dosage of 500mg for patients with ‘mild to severe osteoarthritis’.
Accordingly, the National Evidence-based Health Care Collaborating Agency re-evaluated the current basis on the clinical effect of glucosamine with follow-up research on the existing report, 'the effect of glucosamine and chondroitin in osteoarthritis (National Evidence-based Health Care Collaborating Agency, 2009).
Research Method
This Study was carried out in prompt systematic review method through RAPID, which is a prompt-based evaluation program of the National Evidence-based Health Care Collaborating Agency, and review of literatures published after the publication of reports by this Agency in 2009 and the existing reports. The existing reports and literature search database, research strategy, standard for exclusion from selection and format of data extraction followed those of the existing reports, and search was completed over the period of August 22~23, 2011 by using Ovidmedline, Embase, Cochrane library(including Cochrane systematic reviews database, DARE, NHS EED, HTA database), CRD database, Pubmed, CCTR and Koreamed for review of existing literatures. Random clinical trial research that reported the clinical effectiveness of glucosamine in osteoarthritis and systematic review were used as the selection standard. The process of literature selection, extraction of data and assessment of quality was discussed and agreed upon after having been carried out independently by 2 researchers.
Research Results
Although there were 2 cases of random clinical trial researches (Chopra et al, 2011; Sawitzke 2011) that were added after the existing report, only 1 case (Sawitzke et al, 2011) was added it was not possible to extract the standard deviation at the time of completion of follow-up since 1 case of research (Chopra et al, 2011) presented the resultant values in graph in the meta-analysis that integrated with the existing report. Descriptions of the clinical effect of glucosamine through this Study for each of the medical results are as follows.
Pain reduction effect
- Clinical effect of glucosamine (glucosamine hydrochloride + glucosamine sulfate)
The results of subgroup analysis in accordance with the source of the research grand illustrated that although the integrated estimated value of the glucosamine pain reduction effect was SMD -0.44(95% CI -0.73, -0.16), which was more effective than the placebo, and was different with I2 = 78% in the researches carried out with industrial funding (10 cases), the results of the researches with funding from non-profit organizations (6 cases) displayed no difference in effectiveness from that of placebo with SMD -0.01(95% CI -0.11, 0.09) and I2 = 0%.
As the results of the subgroup analysis in accordance with the methodological quality of execution of research (the adequacy of allocation concealment), the integrated estimated value of researches in which the allocation concealment was carried out appropriately (11 cases) were SMD -0.06(95% CI -0.14, 0.03) and I2=0%, illustrating o effect. On the contrary, researches in which the allocation concealment was not carried out appropriately (7 cases) had SMD -1.05(95% CI -1.65, -0.46), making it effective in comparison to placebo. However, it was different with I2 = 87%.
- Clinical effect of glucosamine sulfate
The results of analysis only of the clinical effects of glucosamine sulfate is the same as the results of the existing reports since there is no literature that has been added after the existing reports, and are as follows.
The results of subgroup analysis in accordance with the source of the research grand illustrated that although the integrated estimated value of the researches carried out with industrial funding (9 cases) was SMD -0.51(95% CI -0.84, -0.18), making it more effective in comparison to placebo, it displayed difference with I2 = 80.7%. The results of the researches with funding from non-profit organizations (3 cases) displayed no difference in effectiveness from that of placebo with SMD 0.01(95% CI -0.18, 0.19) and I2 = 0%.
As the results of the subgroup analysis in accordance with the methodological quality of execution of research (the adequacy of allocation concealment), the integrated estimated value of pain reduction effect in researches in which the allocation concealment was carried out appropriately (7 cases) was SMD -0.08(95% CI -0.20, 0.04) and I2 = 0%, illustrating no effect. On the contrary, researches in which the allocation concealment was not carried out appropriately (6 cases) had SMD -1.21(95% CI -1.91, -0.50), making it effective in comparison to placebo. However, it was different with I2 = 88.5%.
Effect on improvement of joint function
- Clinical effect of glucosamine (glucosamine hydrochloride +glucosamine sulfate)
The results of subgroup analysis in accordance with the source of the research grand illustrated that although the integrated estimated value of the effect of glucosamine on improvement of joint function in the researches carried out with industrial funding (9 cases) was SMD -0.17(95% CI -0.29, -0.06) and I2 = 6%, making it more effective in comparison to placebo, the results of the researches with
funding from non-profit organizations (5 cases) displayed no difference in effectiveness from that of placebo with SMD -0.06(95% CI -0.17, 0.04) and I2 = 0%.
As the results of the subgroup analysis in accordance with the methodological quality of execution of research (the adequacy of allocation concealment), the integrated estimated value of the effect on improvement of joint function in researches in which the allocation concealment was carried out appropriately (11 cases) was SMD
-0.10(95% CI -0.18, -0.01) and I2 = 0%, thereby illustrating very slight effect statistically. It is deemed to lack any meaningful clinical significance since effective size of more than 0.5 clinically is considered to be significant. In addition, researches in which the allocation concealment was not carried out appropriately (7 cases) had SMD -0.50(95% CI -0.91, -0.09), making it effective in improvement of function in comparison to placebo. However, it was different with I2 = 82%
- Clinical effect of glucosamine sulfate
The results of analysis only of the clinical effects of glucosamine sulfate is the same as the results of the existing reports since there is no literature that has been added after the existing reports, and are as follows.
The results of subgroup analysis in accordance with the source of the research grand illustrated that the integrated estimated value of the researches carried out with industrial funding (8 cases) was SMD -0.18(95% CI -0.30, -0.07), making it more effective in comparison to placebo, and I2 = 16.7%. The results of the researches with funding from non-profit organizations (2 cases) displayed no difference in effectiveness from that of placebo with SMD -0.04(95% CI -0.24, 0.17) and I2 = 0%.
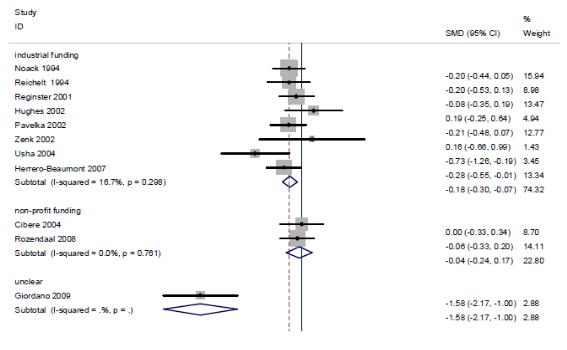
As the results of the subgroup analysis in accordance with the methodological quality of execution of research (the adequacy of allocation concealment), the integrated estimated value of pain reduction effect in researches in which the allocation concealment was carried out appropriately (7 cases) was SMD -0.10(95% CI -0.22, 0.02) and I2 = 0% illustrating no difference in effectiveness from those of placebo. On the contrary, researches in which the allocation concealment was not carried out appropriately (4 cases) had SMD of -0.62(95% CI -1.14, -0.10), making it effective in improvement of function in comparison to placebo. However, it was different with I2 = 85.9%.
Conclusion
Although the results of analysis of the clinical effect of glucosamine in osteoarthritis through the existing reports and additional literature were found to be effective in pain reduction and improvement of joint function in comparison to those of the placebo, there was no consistency in the results. In the subgroup analysis, the researches that were not funded by the manufacturers of glucosamine didn't effect ineffective in pain reduction and improvement of function. In addition, glucosamine has no pain reduction effect and statistically very minor effect in improvement of joint function (SMD -0.10(95% CI -0.18, -0.01) I2=0%) in the researches in which the allocation concealment was appropriate in that the researcher had no clues as to which treatment group the subjects of the research belong to at the beginning of the research. Since the effect size was less than 0.5, making it difficult to deem the results as being clinically significant effective.
In particular, in the case of glucosamine sulfate, which has been approved as medical drug and for which payment of health insurance is acknowledged in Korea, the results of review of the clinical effect found no added literature other than the existing reports. Accordingly, citation of the results of the existing reports without alteration is as follows. Glucosamine sulfate, when compared with the placebo, had
no consistent difference in effectiveness on pain reduction and improvement of joint function in the researches that were not funded by the manufacturers and in researches for which allocation concealment was carried out appropriately.
Since this Study report was reviewed rapidly within short period of time through the RAPID program of the National Evidence-based Health Care Collaborating Agency, it may be somewhat inadequate in terms of its strictness. Accordingly, the risk of being skewed as the result cannot be ruled out. If needed in the future, this aspect could be supplemented by completely systematic review or systematic review that utilizes the existing systematic review.
Recommendation
This Study integrated the present literatures by re-evaluating the clinical effect of glucosamine, which continues to be disputed, and found that glucosamine has no clinically significant effect in pain reduction and improvement of joint function in researches with appropriate allocation concealment and no research funding by
manufacturers.
Although glucosamine is being recognized not only as functional health food but also as medical drug for which health insurance payment is being made in Korea, only Korea, Taiwan and Thailand are the countries in the world that acknowledge payment of health insurance for usage of glucosamine. This could impart inappropriate effect on the health insurance finance, and it is deemed that additional policy determination on whether health insurance finance should be provided for the clinically insufficient effectiveness is needed.
The research results from neutral research institutions without interest relationship with the relevant issues could be the foundation for the objective basis for such policy determination. In the case of overseas countries, it has been stipulate that the HTA evaluation results of NICE of England must be reflected onto the National Health
Services (NHS) within 3 months. In addition, similar institutions in Germany, Australia, Canada, Thailand and Sweden, etc also has the mechanism through which the research results are being fully reflected systematically (refer to the appendix).
Although the research related to the effectiveness of glucosamine that was announced by the National Evidence-based Health Care Collaborating Agency in 2009 was carried out for this purpose, the outcome of the research was not linked to the administrative aspect of the Agency. Although it did cause reduction in the revenue from sales of glucosamine in the market, it did not have sufficient effect on the policy decision such as cessation of sales of and payment of health insurance for administration of glucosamine. Therefore, it is deemed that systematic mechanism for stipulating of the research results of such neutral institutions on to policies in the future is necessary.

 · 저작자표시
· 저작자표시 · 출처표시
· 출처표시
































 한국보건의료연구원
한국보건의료연구원